At Kimia Pharmaceuticals, we are focused on medicine development in key cancer areas where there are patient needs. Our goal is to bring much needed new treatments to people with cancer.
Chronic Myeloid Leukemia and Gastrointestinal Stromal Tumors (GIST)
Our Generic kinase inhibitors, currently are widely used in clinic al treatments, e.g., imatinib
and Sunitinib,produce a more favorable outcome compared to conventional cytotoxic therapy.
These kinase inhibitors have achieved a significant increase in patient survival rate in chronic
myeloid leukemia (CML) and gastrointestinal stromal tumors (GIST), thus translating basic
molecular research into effective patient treatment.
Gastrointestinal stromal tumor (GIST) is the most common mesenchymal tumor of the
gastrointestinal tract. In advanced setting and after progression to imatinib, the multi-targeted
receptor tyrosine kinase inhibitor sunitinib has clearly demonstrated a clinical benefit in terms
of response rate and progression-free survival with an acceptable toxicity profile.
Medicines we are manufacturing target different tyrosine kinase members such as EGFR, VEGFRs,
Kit, PDGFRs, ABL, SRC , all providing improved clinical outcome and patient health status.
we are manufacturing high-quality generic TKIs in the following classification:
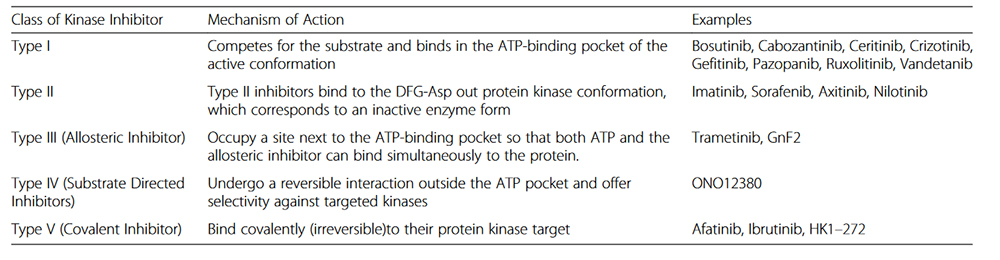
Our products, Erlotinib is among type one EGFR mutated inhibitors which according to NCCN clinical practice guidelines in oncology is included in the first-line treatment of metastatic non-small cell cancer.
This small molecules increase overall survival in real-world clinical practice and also the quality of life in NSCLC patients.Erlotinib, has been shown to improve PFS compared with chemotherapy when given as first-line treatment for Asian patients with NSCLCwith activating EGFR mutations.Our post-marketing experiences show Erlotinib is well tolerated in Iranian patients and has safety profiles equal to its branded one.
In order to increase medicine access for lung cancer patients we recently developed Gefitinib, another first line treatment option for NSCLC which is also an EGFR inhibitor. Patients who progress on Erlotinib now can switch to Gefitinib which has similar safety profile comparing to Erlotinib. Its worth mentioning Some physicians may prefer Gefitinib as first line treatment.